



Research in our lab spans the areas of microfluidics, lab-on-chip, bioMEMS, and bioelectronics for medical applications. The common theme in our projects is instrumentation for obtaining diagnostic data from biological systems, from the level of molecules and cells (for example, digital assays) to the level of the individual (for example, wearable sensors). We are motivated by the premise that more data improves our understanding of life, and can help us make better decisions about our health and environment. Current projects involve two thrusts:
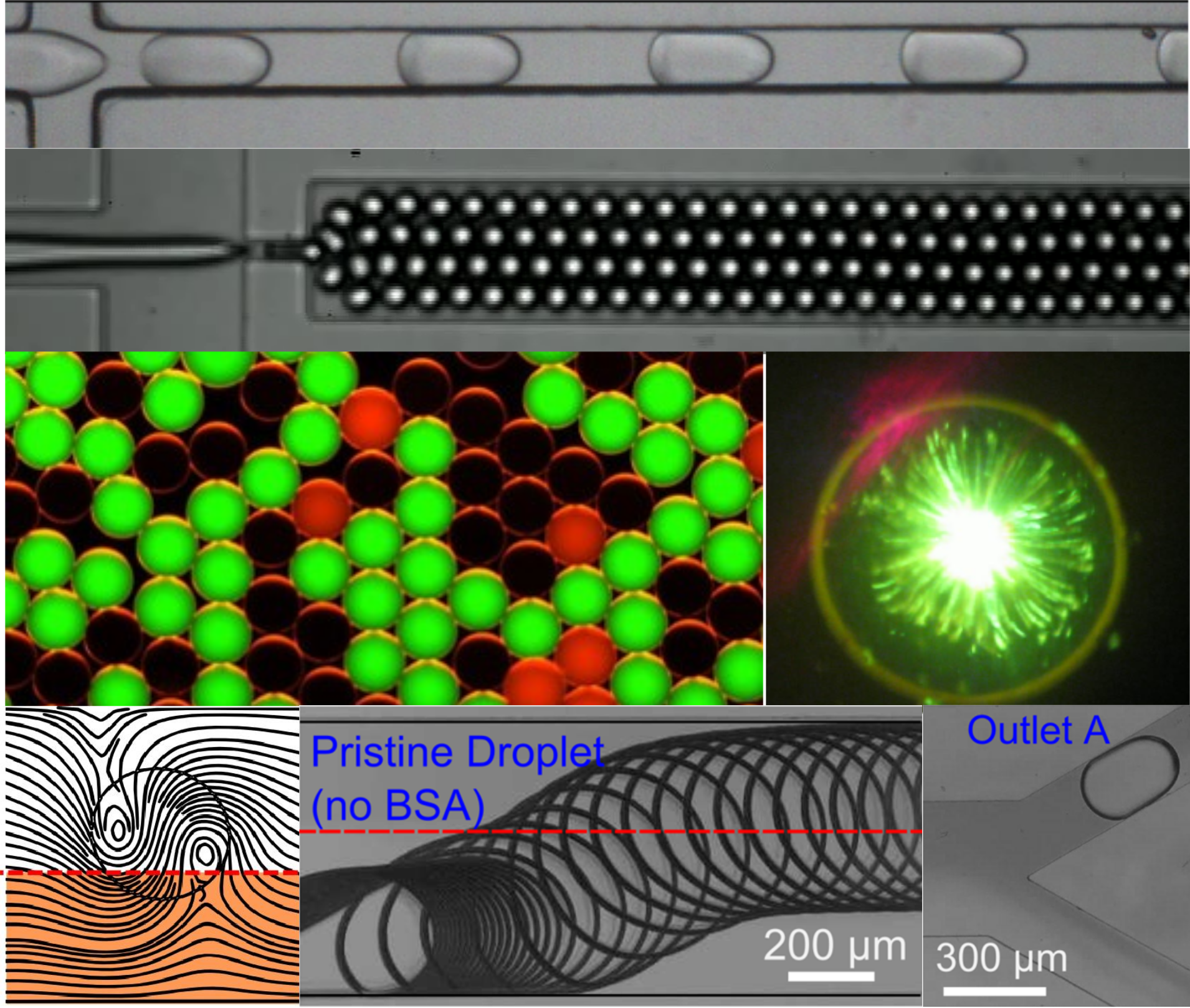
Modern biological research relies on high-throughput screening (HTS) instruments which can perform assays involving large numbers biomolecules or cells. These instruments must be able achieve high speed, low cost, and minimal reagent consumption. Particularly well suited for HTS is a class of microfluidic devices which utilizes water-in-oil droplets as chemical reaction containers. With typical dimensions of <100 micrometers and with volumes on the order of nanoliters to femtoliters, droplet systems have the potential to manage large libraries of chemical reactions while consuming minimal amounts of reagents. Droplet systems have been widely used for modern methods of digital PCR, single cell assays, chemical fractionation, and more. Our work in this area focuses on the physics and applications of multiphase flow and interfacial phenomena, the manipulation and storage of droplets, and label-free biodetection. Applications of these devices include digital assays, including single molecule, single cell, and single organism screening.

Microelectronics and microsensors have become pervasive in the modern age, creating a significant opportunity for their use in portable, wearable bioinstruments for distributed and home health monitoring. We are motivated by the use of these devices to help manage chronic disease, which accounts for >65% of US health care costs. Our work in this area takes a translational approach, focused on developing systems to serve needs in healthcare as well as environmental applications. Current projects include ultraminiature wearable biosensors for fitness and stress monitoring, portable biodetection systems for detecting invasive species in foreign vessels entering the Great Lakes, and multiplexed electronics for portable flow cytometry systems.








A. Vedhanayagam
In this project we perform rapid image-based flow cytometry utilizing GPU-Accelerated computer vision and machine learning. Our systems are designed to meet the throughput demands of liquid biopsy and environmental screening.

P. Weerappuli
We developed a scalable droplet random access memory (DRAM), a microfluidic device in which droplets and single cells can be addressably stored and released.

M. Rezaei, A. Mandhare, A. Sunkari, N. Raitu, D. Ulmer, O. Sufi, A. Karaali
TRACE is a novel, earlobe-mounted heart rate monitor which provides beat-to-beat accuracy in fitness and health monitoring applications, based on our patented heart rate sensing method.

A. Fatima
We are developing a novel, label-free, inline chemical detector based on the surfactant retardation effect, first discovered in the 1960s. By amplifying this effect in microfluidic channels, we can use it to detect proteins and surface-active molecules at ng/mL concentration.

A. Akram, S. Noman, R. Javid, J. Ram
We are developing systems to detect live organisms in ballast water, for preventing invasive species in the Great Lakes.

A. Vedhanayagam
Droplet Morphometry and Velocimetry (DMV) is a video processing software for tracking the droplet size, velocity, trajectory, and other parameters. It is available for free for academic researchers, and is used worldwide.

G.K. Kurup
Tensiophoresis is the chemomechanical force on a droplet, which can be used to sort droplets by size and chemical composition.

G.K. Kurup, A. Doshi
Optofluidic tweezers utilizes balanced thermocapillary flows to trap and manipulate droplets using a focused laser beam. OFT have forces more than 100,000 times greater than traditional optical tweezers.

S. Hamed, P. Sehgal, M. Utomo
Droplet fractionation encapsulates chemically separated compounds into droplets, enabling nL-pL fractions to be collected without physical containers.

G.K. Kurup
This project concentrates particles within droplets by exploiting microvortex flow within the droplet.

V. Trivedi, A. Doshi, P. Sehgal
We demonstrate that droplet generation, merging, and analysis can be carried out in commercial laboratory tubing.

T. Mertiri, M. Chen
We developed 96-well plates with the ability to control light intensity in each well. These have been used for screening photosynthetic algae and more recently for optogenetic screening.
Please Note: Downloads are for personal use only, and may not be distributed without permission of the publisher.

BioMEMS and BioInstrumentation is an interdisciplinary graduate level course is open to students in engineering, chemistry, physics, and life sciences. It will cover the fundamentals of biomedical micro and nanosystems, with a focus on lab-on-a-chip technologies. Students will learn about the miniaturization of analytical systems used in biology, chemistry, and medicine. Topics of interest to engineering and physics students include: micro and nanofabrication techniques, the physics of microfluidic flow and other microscale phenomena, and computational modeling techniques. Students in life science and chemistry will learn emerging analytical techniques, molecular/particle/cell separations on chip, single cell assays, and detection methods.
Lecture Recordings
ECE 4570 is a senior level undergraduate course covering aspects of electrical properties of semiconductors, the physical electronics of P-N junction, bipolar, field effect transistors, and device fabrication technology essential to understanding semiconductor active devices and integrated circuits. Introduction to the behavior of semiconductor and electronics devices. Students also take part in a semiconductor fabrication lab, where they learn about the microfabrication processes used to manufacture and test semiconductor devices including resistors, diodes, and MOSFETs.
 Nanotechology is quickly becoming a pervasive and fundamental component of science and technology. The microfluidics laboratory is collaborating with the Detroit Science Center, the city's premiere source for K-12 science education, to develop modules for educating and inspiring future scientists and engineers in the micro and nanoscale worlds.
Nanotechology is quickly becoming a pervasive and fundamental component of science and technology. The microfluidics laboratory is collaborating with the Detroit Science Center, the city's premiere source for K-12 science education, to develop modules for educating and inspiring future scientists and engineers in the micro and nanoscale worlds.
 Sponsored by the National Science Foundation's Michigan Louis Stoke Alliance Minority Participation (MI-LSAMP) program, SURA is an effort designed by Wayne State's MI-LSAMP Work Group to provide summer research opportunities primarily to first and second year undergraduate students at Wayne State and other partner institutions, including Michigan State University, University of Michigan-Ann Arbor, and Western Michigan University. MI-LSAMP aims to significantly increase the number of minority students earning baccalaureate degrees each year in Science, Technology, Engineering and Mathematical (STEM) fields and prepare them for entry into graduate programs.
Sponsored by the National Science Foundation's Michigan Louis Stoke Alliance Minority Participation (MI-LSAMP) program, SURA is an effort designed by Wayne State's MI-LSAMP Work Group to provide summer research opportunities primarily to first and second year undergraduate students at Wayne State and other partner institutions, including Michigan State University, University of Michigan-Ann Arbor, and Western Michigan University. MI-LSAMP aims to significantly increase the number of minority students earning baccalaureate degrees each year in Science, Technology, Engineering and Mathematical (STEM) fields and prepare them for entry into graduate programs.
